上流の強化の進化:PATによる灌流プロセスの最適化
上流の強化の進化:PATによる灌流プロセスの最適化
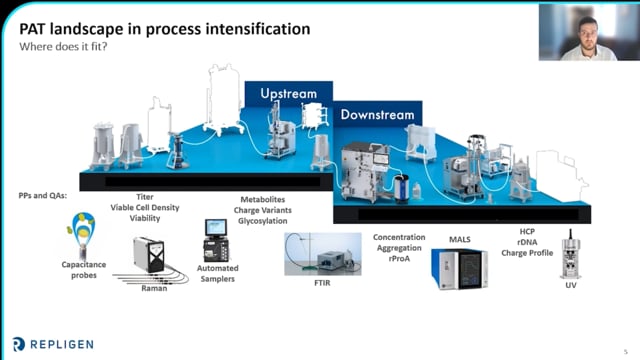
Process analytical technology (PAT) in bioprocessing has improved manufacturing control while reducing variability and costs in process development and manufacturing. Leveraging PAT with process intensification tools drives robust, efficient, and scalable platforms. In this webinar, Bristol Myers Squibb scientists describe application of PAT to upstream intensification towards developing a mAb N-1 platform process with accelerated timelines. The optimal cell-specific perfusion rate and inoculation density were determined within 2- 3 experiments. The PAT landscape in upstream intensification, the benefits of this approach, and the scale-up from benchtop to manufacturing will be discussed.
Discover More Webinars
01
02
03
04
05
01
/
05