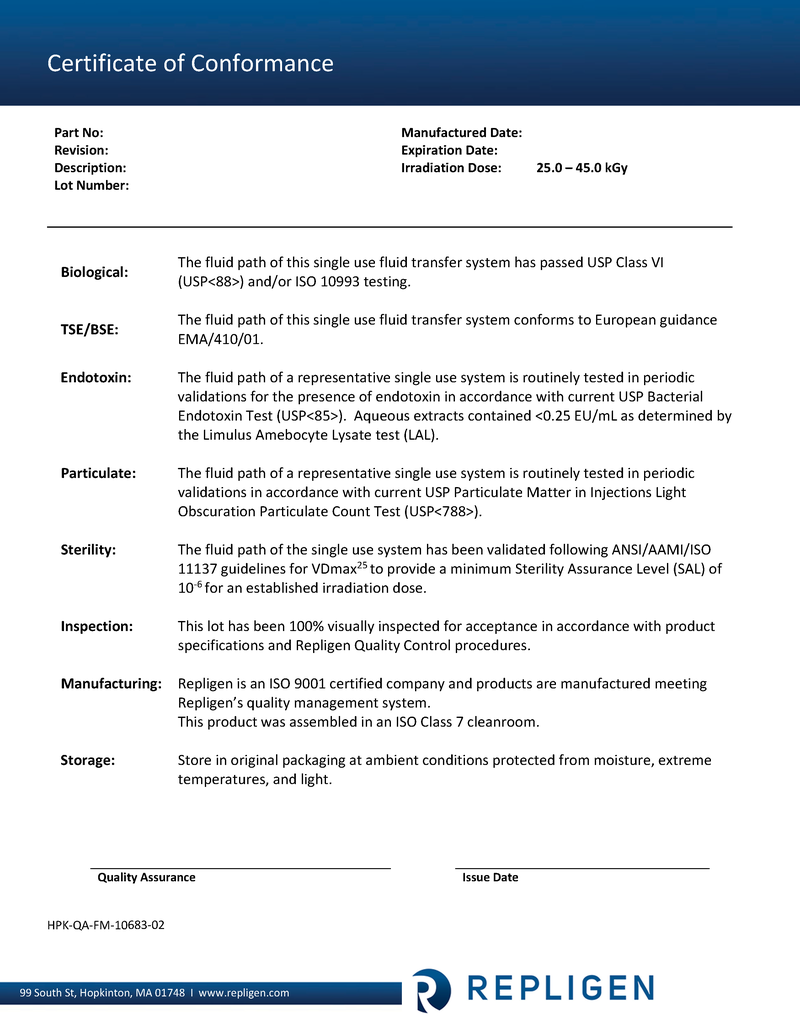
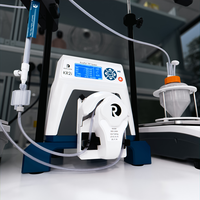
ProConnex® Flow Paths for Systems
Expertly designed plug-and-play flow path
ProConnex® Flow Paths for TFF and Chromatography processes are single-use and engineered for optimum performance, reproducibility and time savings. System specific flow paths provide speed and reliability. Configured flow paths prioritize process requirements and sterility.
Configurable System Flow Paths
Engineered for specific applications and requirements
NEW ProConnex® TFF Self Assembly Kits
Available today for KrosFlo® KR2i System
- Simplify your process: One flexible assembly kit has everything you need — just add a bottle, filter and go.
- Ensure quality and traceability: Single CoQ and MFG lot traceability for consistent, reliable results every time.
- Built for your KrosFlo® system: Designed with Repligen expertise and for seamless integration with Spectrum® Hollow Fiber. All from a single supplier.
| See datasheet | 専門家に相談する |
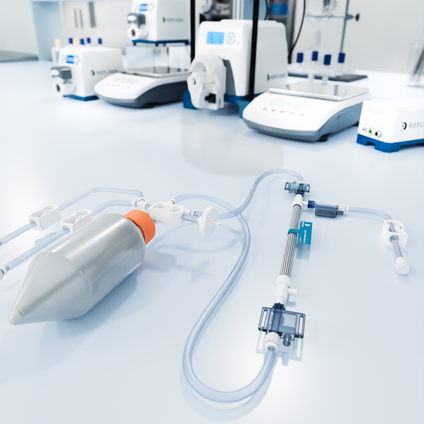
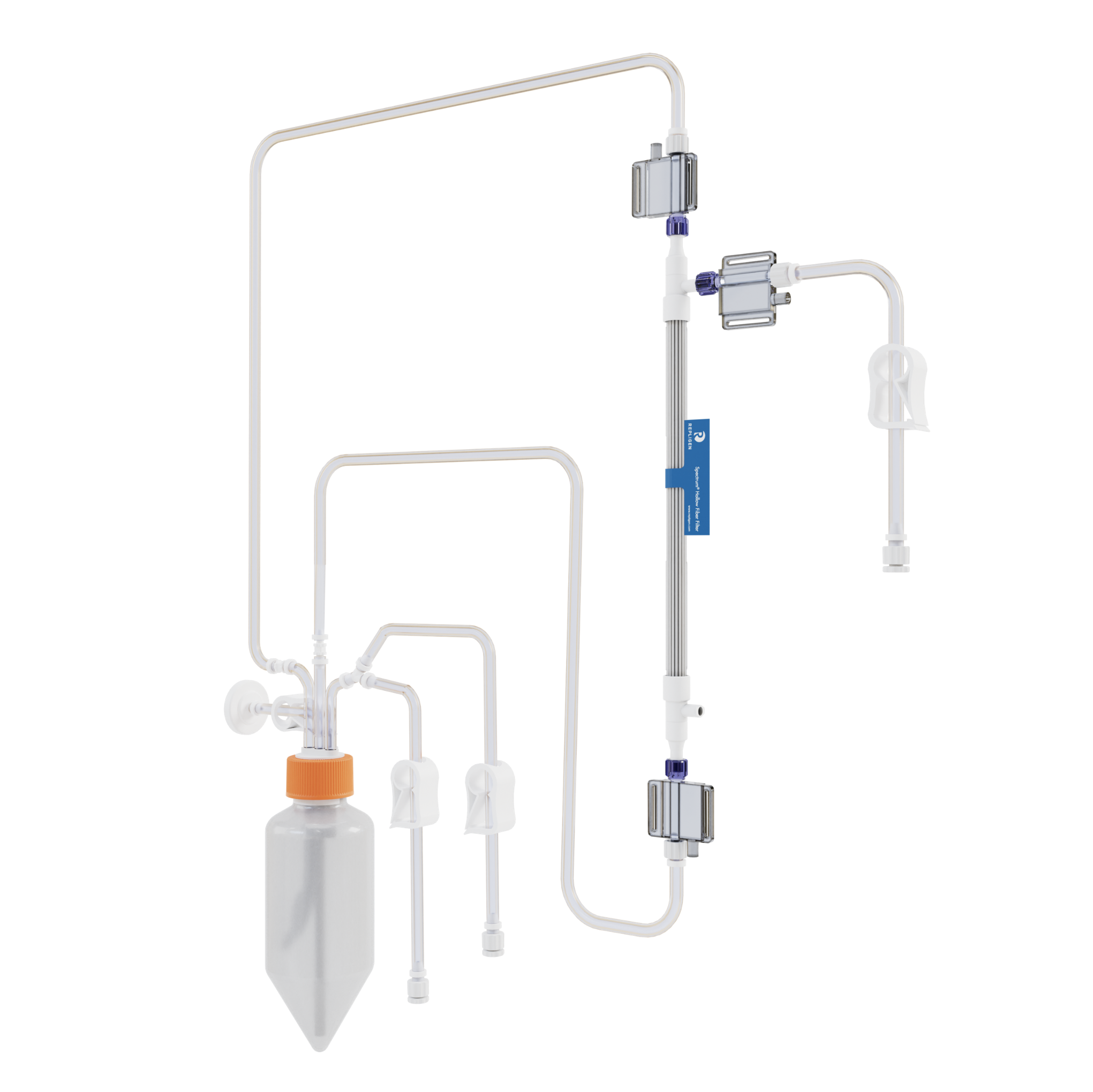
KrosFlo KR2i and KrosFlo KMPi Flow Paths
The convenience of off-the-shelf flow paths designed specifically to run Spectrum® Hollow Fiber or TangenX® flat sheet cassettes on the KrosFlo KR2i and KMPi systems. Filter sold separately.
|
Design Your Own Configure a KrosFlo KR2i or KrosFlo KMPi flow path to meet specific process requirements. Includes quality documentation and free consultation. |
 |
|
Order Parts You don’t mind the work of assembling your own flow path. Our selection of connectors, pressure sensors, tubing and vessels could be the solution for you. |
 |
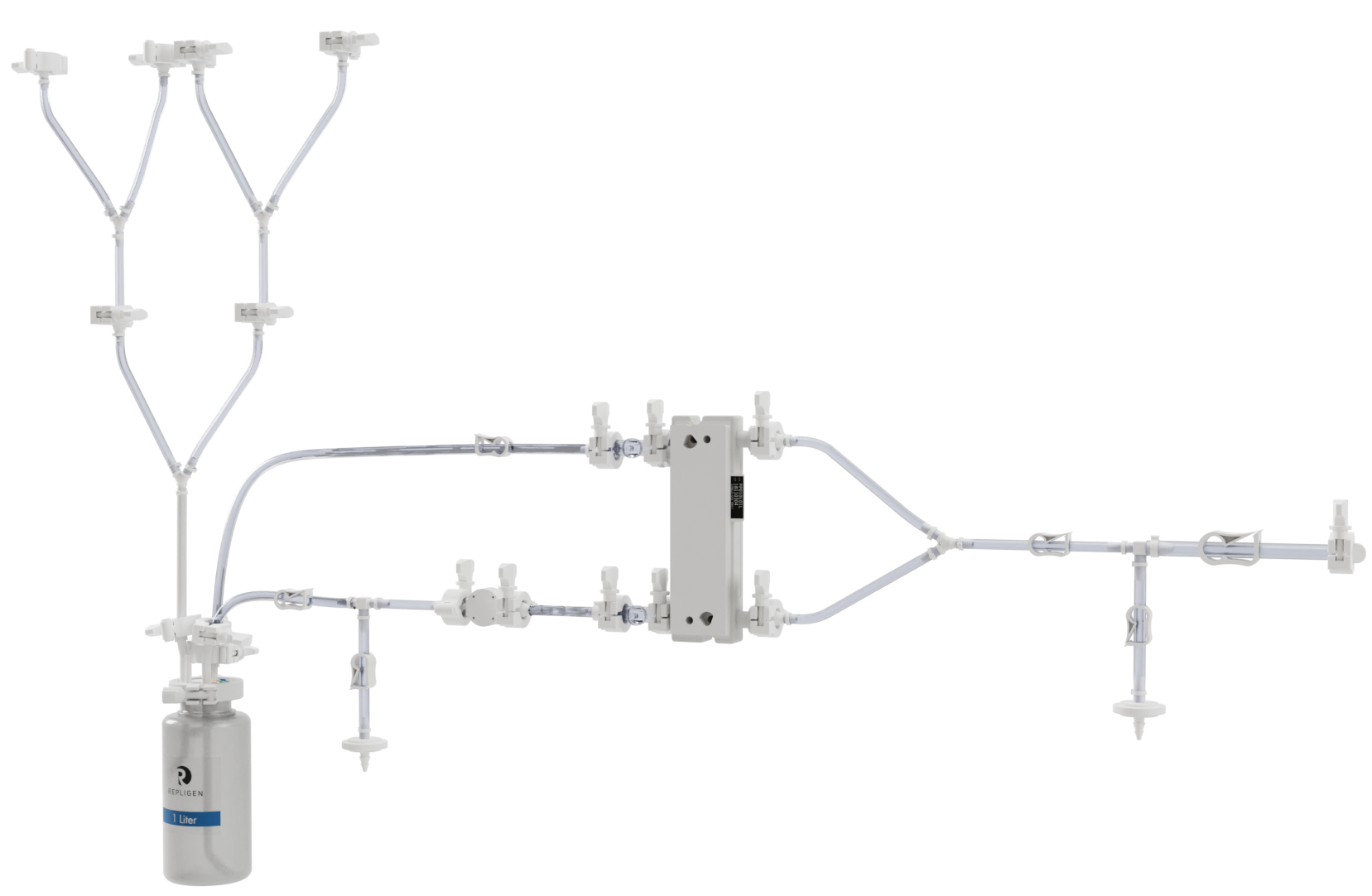
KrosFlo FS-15 and FS-500 Flow Paths
Off-the-shelf flow paths offer convenience, reliability and quality documentation for flat sheet cassettes run on FS systems. No more cutting tubing, component searching, documentation and inconsistent setup. Filter sold separately.

|
Design Your Own Configure a FS flow path to meet specific process requirements. Includes quality documentation and free consultations. |
 |
|
Order Parts You don’t mind the work of assembling your own flow path. Our selection of connectors, pressure sensors, tubing and vessels could be the solution for you. |
 |
KrosFlo TFDF Lab Flow Paths
Off-the shelf, single-use, gamma-irradiated and closed TFDF Lab Flow Paths arrive dry, free of sodium hydroxide, glycerin, preservative and ethanol, for a zero flush start state.

Custom KrosFlo TFDF Pilot and Process Flow Paths
When scaling up, configure a flow path to meet your process requirements for your pilot and process KrosFlo TFDF Systems. All KrosFlo TFDF flow paths are single-use, gamma-irradiated and arrive dry, free of sodium hydroxide, glycerin, preservative and ethanol, for a zero flush start state.

KrosFlo KTF Flow Paths
Configure a flow path for either Spectrum Hollow Fibers or TangenX flat sheet cassettes that matches the design of your KrosFlo system.

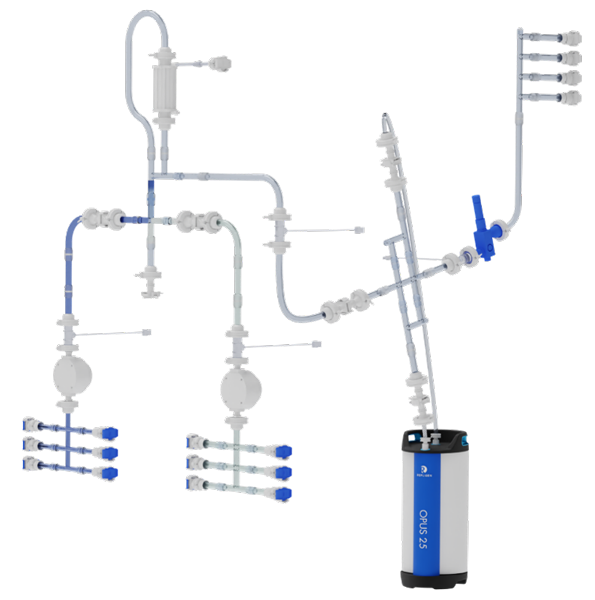
KRM Chromatograhy System Flow Paths
Highly optimized, low hold-up volume flow paths that utilize innovative technologies like overmolded connections and injection molded intersections for optimal fluid dynamics on the advanced KRM Chromatography Systems.
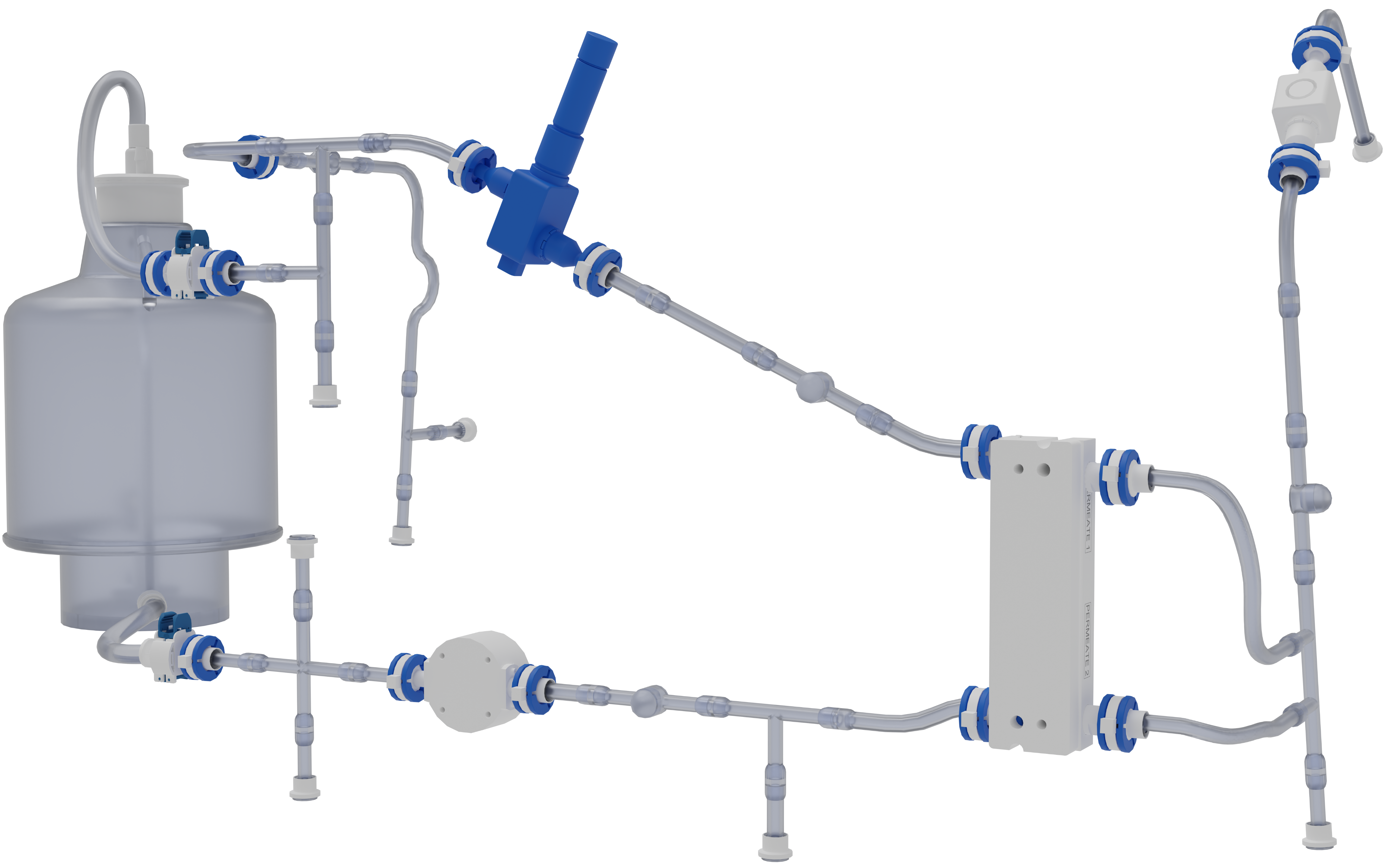
KrosFlo RS TFF System Flow Path
Highly optimized, low hold-up volume flow paths that utilize innovative technologies like overmolded connections and injection molded intersections for optimal fluid dynamics on the highly automated KrosFlo RS Systems.
 SYSTEM FLOW PATHS
SYSTEM FLOW PATHS
Save time, streamline operations and hit the ground running
These off-the-shelf flow path solutions offered by Repligen deliver wins for the customers every step of the way.
.png)
 SYSTEM FLOW PATHS
SYSTEM FLOW PATHS
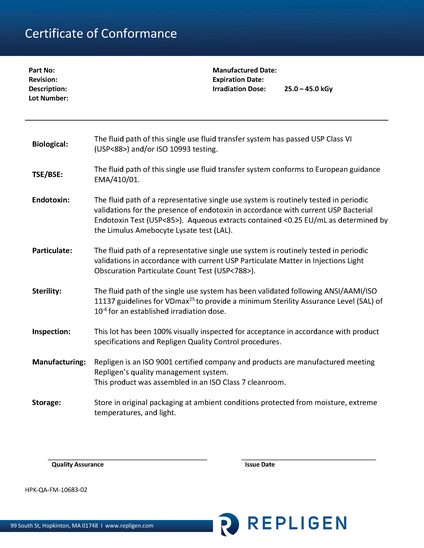
High quality standard to minimize regulatory burden
BIO USP <88> TSE/BSE
- The fluid path of this single use fluid transfer system has passed USP class VI (USP<88>) and EMA/410/01
PARTICULATE USP <788>
- Current USP standard for allowable visible and subvisible particulate for medical infusions an injections
ENDOTOXIN USP <85>
- Current standard for quantification of endotoxin, Aqueous extracts from representatives samples contained <0.25 EU/mL
STERILITY ANSI/AAMI/ISO 11137
- Validated following ANSI/AAMI/ISO 11137 guidelines for VDmax25 to provide a minimum sterility assurance level (SAL) for 10-6 for
 CONFIGURABLE FLOW PATHS
CONFIGURABLE FLOW PATHS
Flow paths engineered to user specifications and requirements
Leverage a library of over 300 individual components and expert support to turn specific process requirements into an optimal flow path in no time.
Get the most out of the process and Repligen system while maintaining sterility and still being plug and play.
These custom configured flow path are supplied fully assembled, complete with accessories and filters.
 CONFIGURABLE FLOW PATHS
CONFIGURABLE FLOW PATHS
Technical consultation from concept to product

With decades of experience, Repligen technical experts match your core technology with the flow paths that suit your application.
Process goals
Core technology, application, facility fit
Process parameters
Feed flow rate, working volume and pressure, integrity test, sterility, scale
Flow path requirements
Aseptic connectors, sensors, vessel, hold-up volume
Flow path specifications
Engineering documents, bill of materials, design lock
High-Capacity, Globalized Manufacturing and Supply Chain
A supply chain that prioritizes best-in-class lead times and 95% on time delivery by designing for robustness, redundancy and risk mitigation. System specific flow paths and certain hollow fiber filters are stocked and available off-the-shelf.

Membrane and module manufacturing
- Multi-lane
- Multi-site
- Dual continent

Assembly and gamma radiation
- Multi-lane, multi-site, and dual-continent assumbly centers
- Multi-cell and multi-site gamma radiation with 2 independant partners

Packaging and logistics
- Continuous improvement programs
- ISTA3A certified packaging process
- Networked and regional distribution
- Sustainability programs
Key Specifications for System Flow Paths
Below are the key specifications for system flow paths. To see specifications for KrosFlo® KTF and RS TFF Systems or the KRM™ Chromatography System, please contact a sales representative.
Manufacturing Centers of Excellence
Repligenは、ISO 9001品質管理システムに基づいて、バイオ医薬品業界向けの製品を開発・製造しています。高品質で安定した、堅牢な製品をタイムリーに提供し、お客様の事業継続性を保証することを重視しています。
Repligen manufacturing sites are located in Massachusetts, California, and New Jersey in the United States and in Sweden, France, The Netherlands, Germany and Estonia.


お客様第一。
サポートは、Repligenという企業の遺伝子に組み込まれています。弊社の目標は、卓越した顧客体験を提供すること、そしてRepligenの製品やサービスの適用や導入を効率よく成功に導くためにサポートすることです。
- Field Application Support
- カスタマーサービス
- フィールドサービスエンジニア