灌流細胞培養
上流の連続バイオプロセシングの基礎となる灌流は、1980年代から施設の使用率を最大化し、プロセスの柔軟性を向上しコストを最小化する目的で実践されてきています。そして今、高度な制御ロジックを備えた新しい細胞保持装置とシングルユース技術により、実装が簡単になっています。

Perfusion cell culture utilizes a cell retention device and continuous media exchange to achieve and maintain high cell densities and viabilities over extended periods of time. The cell retention device retains cells inside the bioreactor, while fresh media is added, and products of interest, waste products and spent (or depleted) media are continuously removed. Fresh media is provided at the same rate that product and spent media are removed from the bioreactor. Hollow fiber-based membrane filters act as the most reliable and commonly used membrane type. Long-term perfusion is typically performed in weeks.
Technology innovations, such as XCell ATF®, solved historical challenges, shifting the paradigm towards perfusion as a key platform for modern upstream process intensification and continuous processing.
Repligen solutions help overcome key challenges in perfusion cell culture, with hands-on process and implementation consultation from global Field Applications Specialists.
Long-term Perfusion Cell Culture Workflow
Click on the workflow link below to find productivity and throughput solutions you can implement today.
Perfusion enabled 10X VCD and 10X product than Fed-Batch
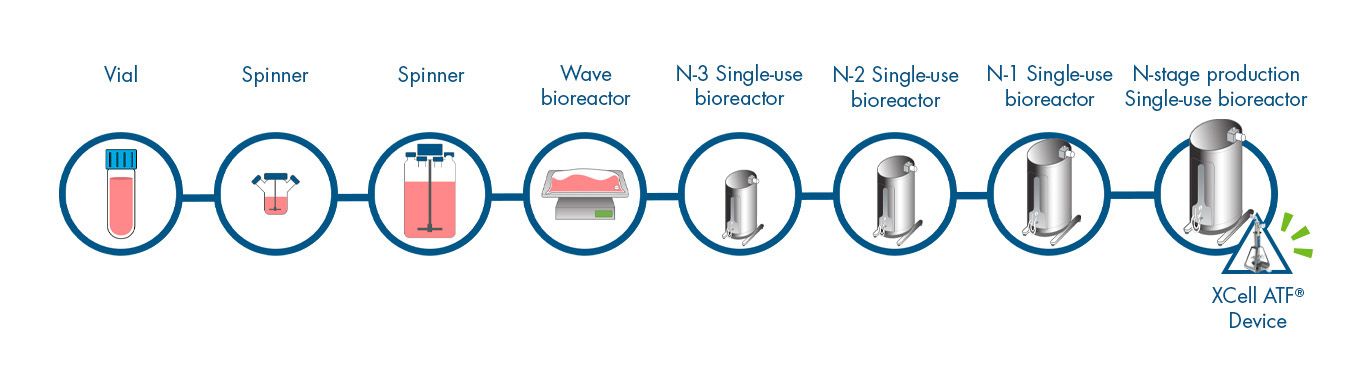
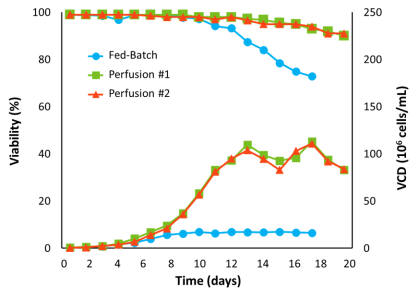
Perfusion achieved 100-130 million viable cell density (VCD) compared to 13 million for Fed-Batch. Perfusion culture viability was maintained above 90% over a period of 17 days. Fed-Batch viability began to drop after day 9 to approximately 70% at days 14-15. Higher VCD and viability of perfusion cultures translated to an increase in total product yield. The perfusion process yielded approximately 30-35 g, while the Fed-Batch process achieved 3 g in approximately 15 days.
Higher cell density, higher viability
- Unoptimized perfusion process achieved up to 10X VCD over optimized Fed-Batch
- Perfusion enabled higher cell growth rates and healthier cultures
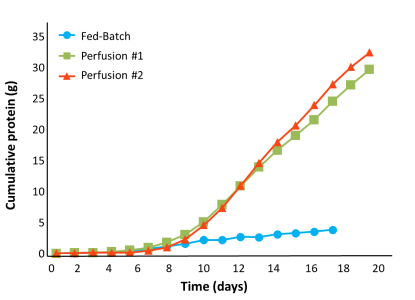
高い生産性
- Higher cell density and viability of perfusion culture enabled 10X yield increase over Fed-Batch

お客様第一。
サポートは、Repligenという企業の遺伝子に組み込まれています。弊社の目標は、卓越した顧客体験を提供すること、そしてRepligenの製品やサービスの適用や導入を効率よく成功に導くためにサポートすることです。
- Field Application Support
- カスタマーサービス
- フィールドサービスエンジニア